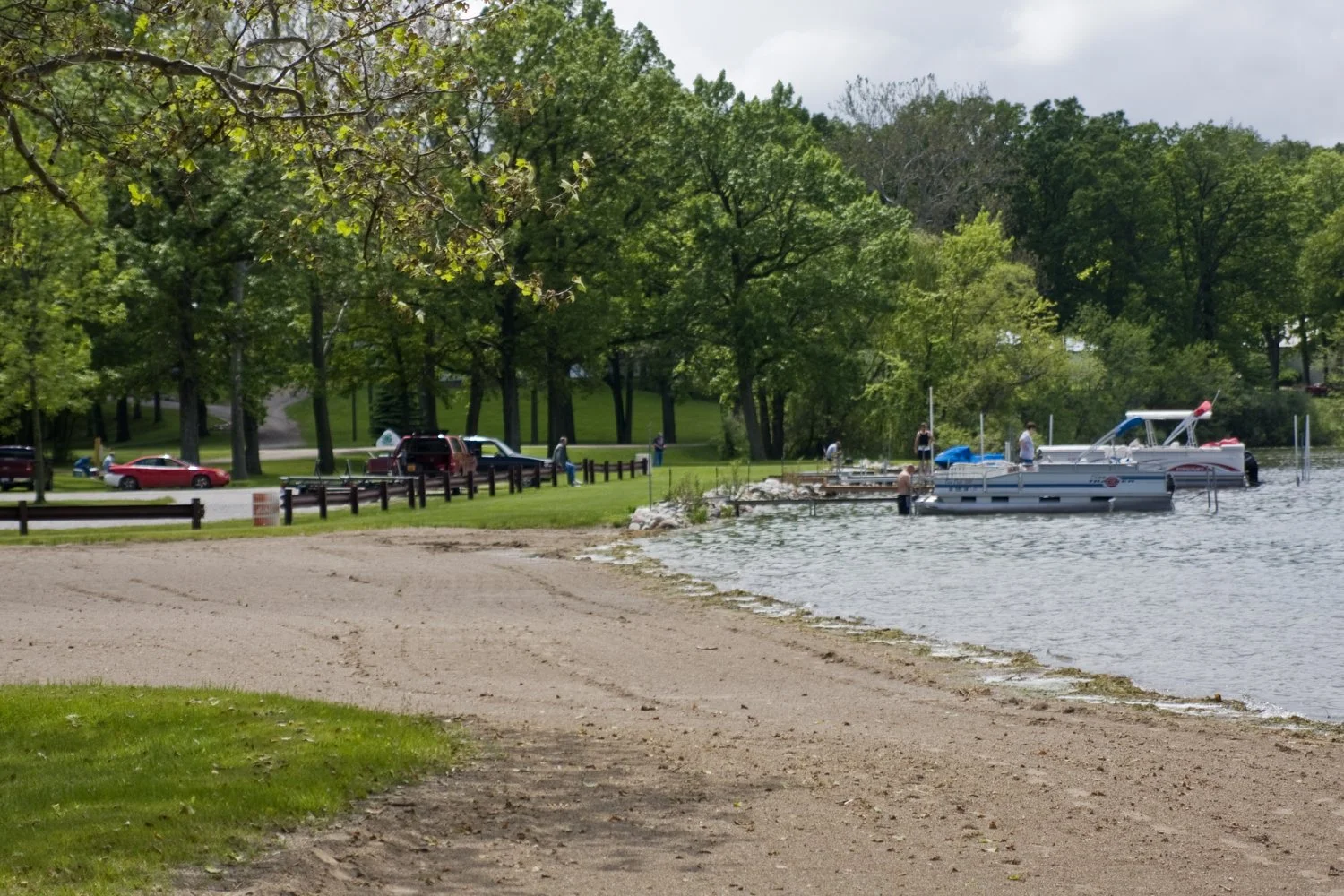Latest News
Steuben County Opens Cooling Centers
(STEUBEN COUNTY) - Steuben County officials are encouraging residents to take precautions as hot and humid conditions settle into the region this week.Steuben County Emergency Management has released a list of cooling centers available to residents seeking relief from the heat.Cooling Centers• YM...
Read MoreRed Cross Urges Blood, Platelet Donations As Summer Shortage Looms
(FORT WAYNE) - The American Red Cross is urging donors to give blood or platelets as donations continue to lag behind seasonal demand.The organization says blood donations are down about 25,000 units nationwide over the past several weeks, raising concerns about maintaining an adequate supply hea...
Read MoreColdwater Utility Fee Lawsuit Faces Key Hearing Thursday
(COLDWATER) - A class-action lawsuit challenging utility fees charged by the City of Coldwater is scheduled for a key court hearing Thursday.The lawsuit was filed by Coldwater resident Jason Mate and alleges the Coldwater Board of Public Utilities has overcharged electric customers by calculating...
Read MoreIndiana Michigan Power Suspends Power Shutoffs During Heat Wave
(FORT WAYNE) - Indiana Michigan Power is temporarily suspending service disconnections for nonpayment as dangerous heat settles across the region.The utility announced that power shutoffs for nonpayment will be suspended Wednesday, June 10, throughout its service territory because of forecast ext...
Read MoreSteuben County EMS Grappling With Overtime Costs Amid Paramedic Shortage
(ANGOLA) - Steuben County EMS is facing rising staffing costs as paramedic vacancies continue to strain the department's budget.During Tuesday's meeting of the Steuben County Council, EMS Administrator Alicis Walsh requested a budget transfer to cover increased part-time staffing expenses.Walsh s...
Read MoreSteuben County Council Approves Nearly $390,000 In Park Improvements
(ANGOLA) - The Steuben County Council has approved nearly $390,000 in improvements at the county park, addressing several long-standing maintenance and infrastructure needs.The funding will come from the county's Major Moves fund and will support a series of projects discussed during Tuesday's co...
Read MoreSteuben Democrats Seek Candidates For Multiple County Offices
(ANGOLA) - The Steuben County Democratic Party is seeking candidates to run for several county offices in the November election.Party leaders announced they are looking to fill positions on the fall ballot and are encouraging interested residents to come forward.Open countywide races include asse...
Read MoreAlligator Owner Defends Operation As Search Continues In Auburn
(AUBURN) - The owner of an alligator that escaped in Auburn last week is pushing back against concerns from neighbors as the search for the reptile continues.Mark Kohlhorst, owner of the educational animal program Mark's Ark, said he discovered the two-foot-long alligator missing Thursday evening...
Read MoreMSP Awaits Toxicology Results In Fatal I-69 Crash Investigation
(COLDWATER) - Michigan State Police say a fatal Interstate 69 crash that killed a Branch County Road Commission employee remains under investigation as authorities await toxicology test results from the driver of a semi-truck involved in the collision.The crash happened June 5 when a semi-truck r...
Read MoreWork Release Program Could Be Eliminated After State Funding Cuts
(ANGOLA) - The work release program serving Steuben and LaGrange counties could be eliminated later this year as local officials prepare for a significant reduction in state funding.Speaking to the Steuben County Council on Tuesday, Northeast Indiana Community Corrections Director Brett Hays said...
Read More